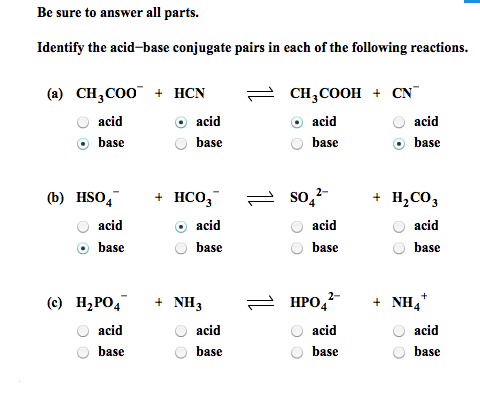Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com

SOLVED: 4) Fill out the conjugate acids/bases in the following table: Conjugate Acid Base Pairs Acid: HF Conjugate base: Base: SOa" Conjugate acid: Base: Cl Conjugate acid: Acid: HCIOa Conjugate base: Acid:
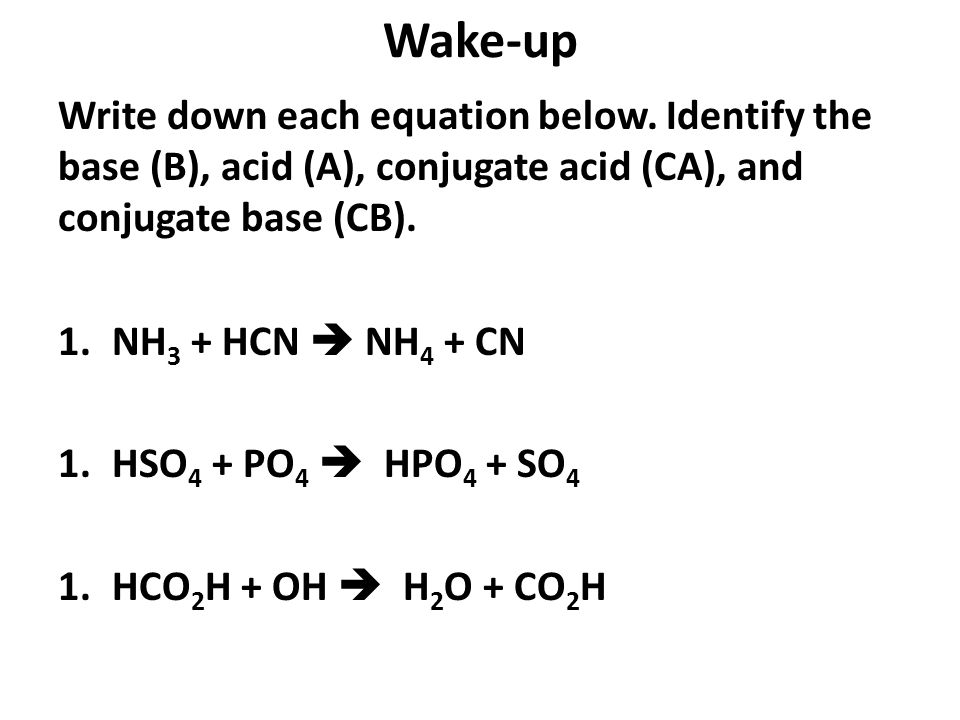
Wake-up Write down each equation below. Identify the base (B), acid (A), conjugate acid (CA), and conjugate base (CB). 1.NH 3 + HCN NH 4 + CN 1.HSO ppt download

Pd-Catalyzed C–N Coupling Reactions Facilitated by Organic Bases: Mechanistic Investigation Leads to Enhanced Reactivity in the Arylation of Weakly Binding Amines | ACS Catalysis